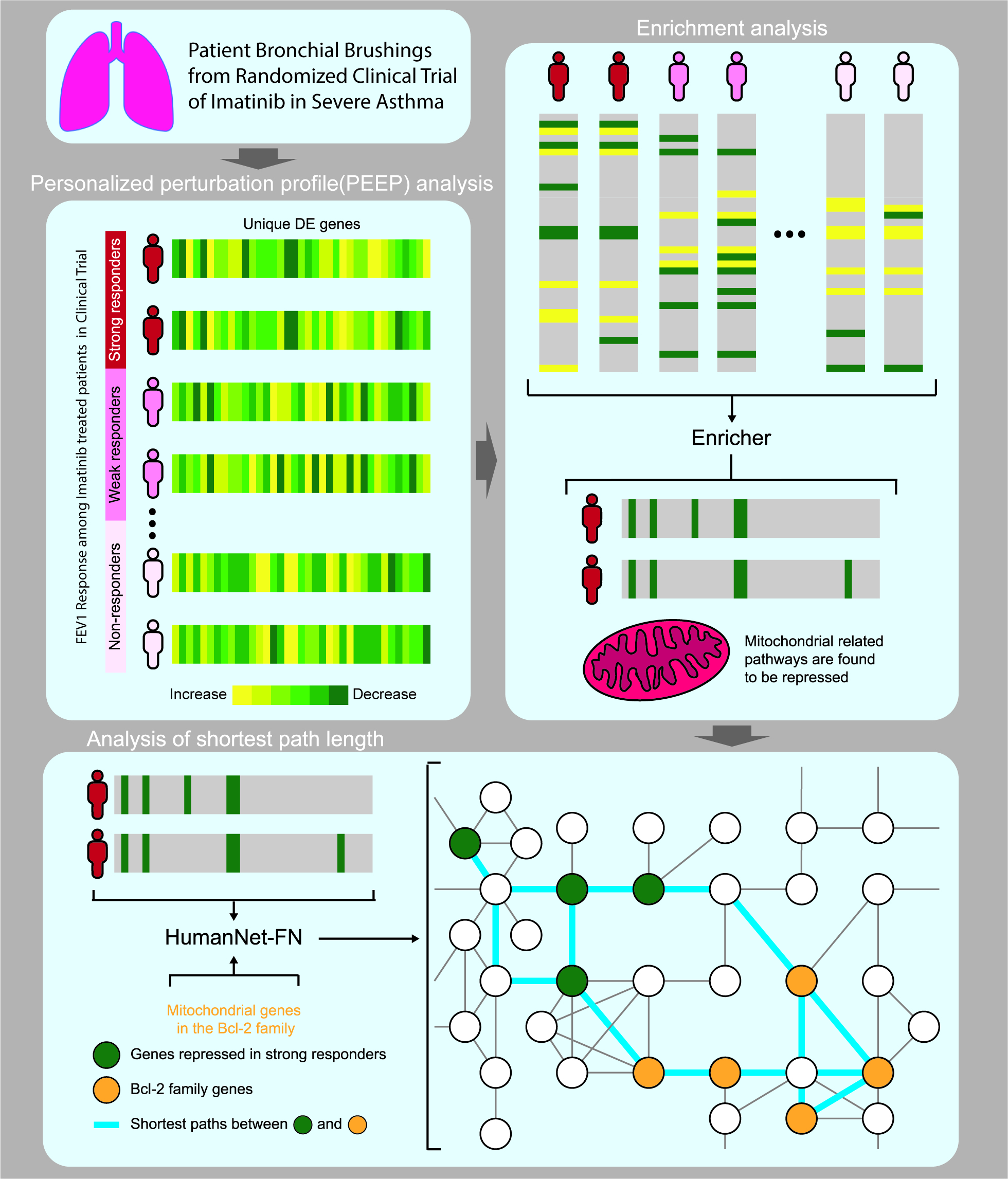
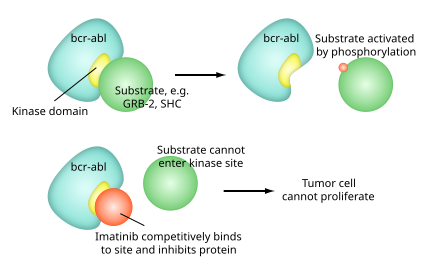
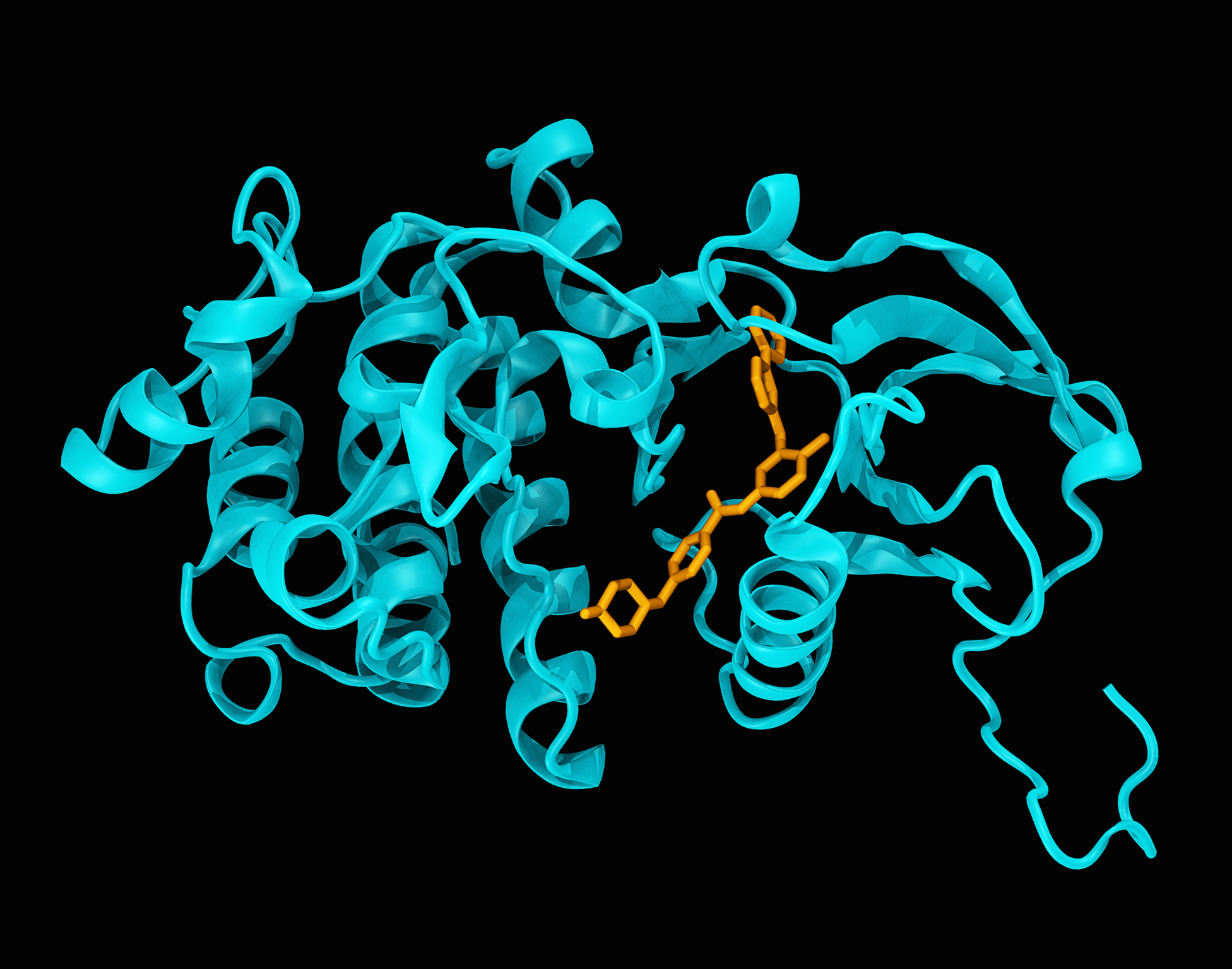
Ponatinib versus imatinib for newly diagnosed chronic myeloid leukaemia: an international, randomised, open-label, phase 3 trial - The Lancet Oncology

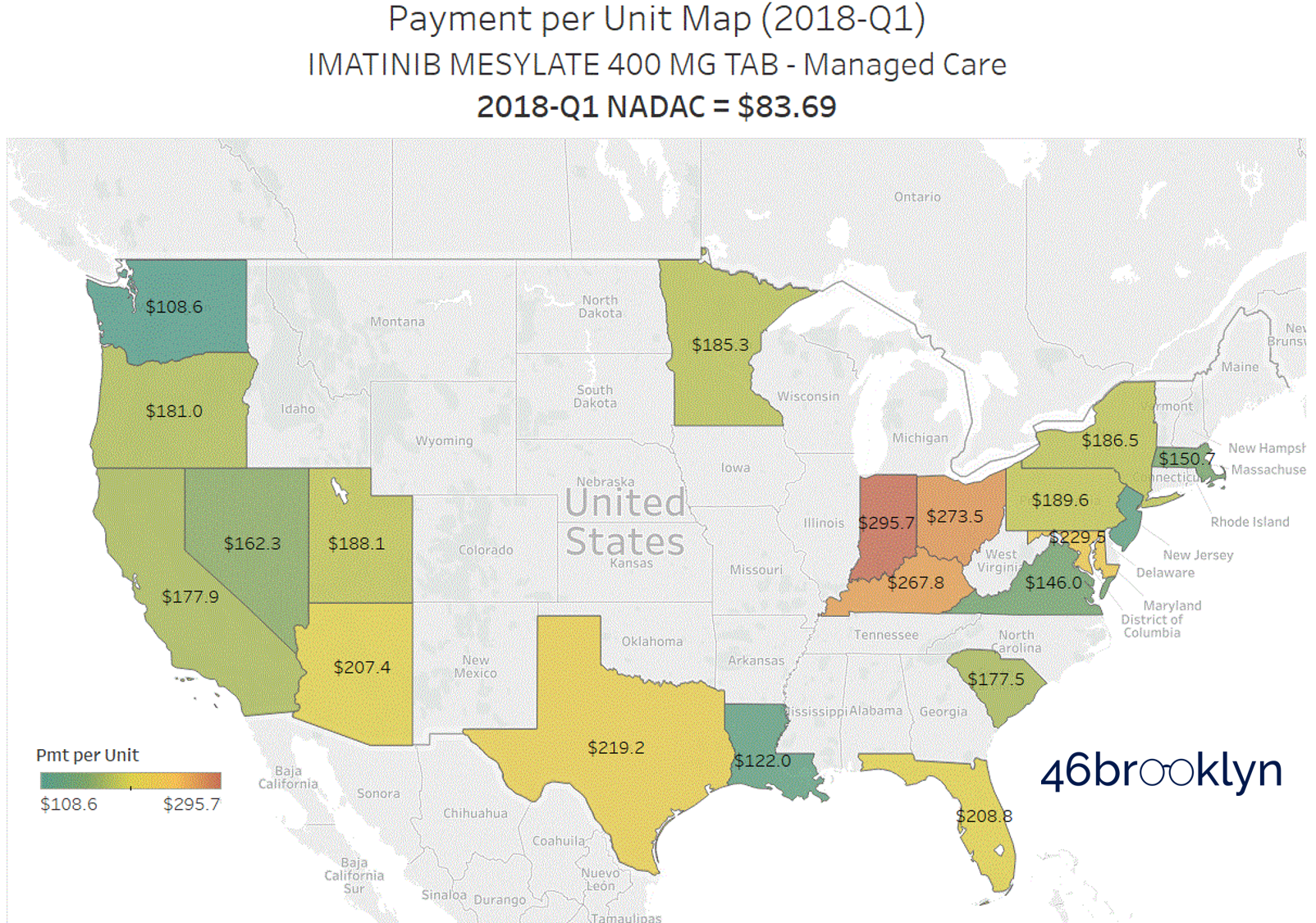
Current evidence on the efficacy and safety of generic imatinib in CML and the impact of generics on health care costs - ScienceDirect

Switching to nilotinib versus imatinib dose escalation in patients with chronic myeloid leukaemia in chronic phase with suboptimal response to imatinib (LASOR): a randomised, open-label trial - The Lancet Haematology

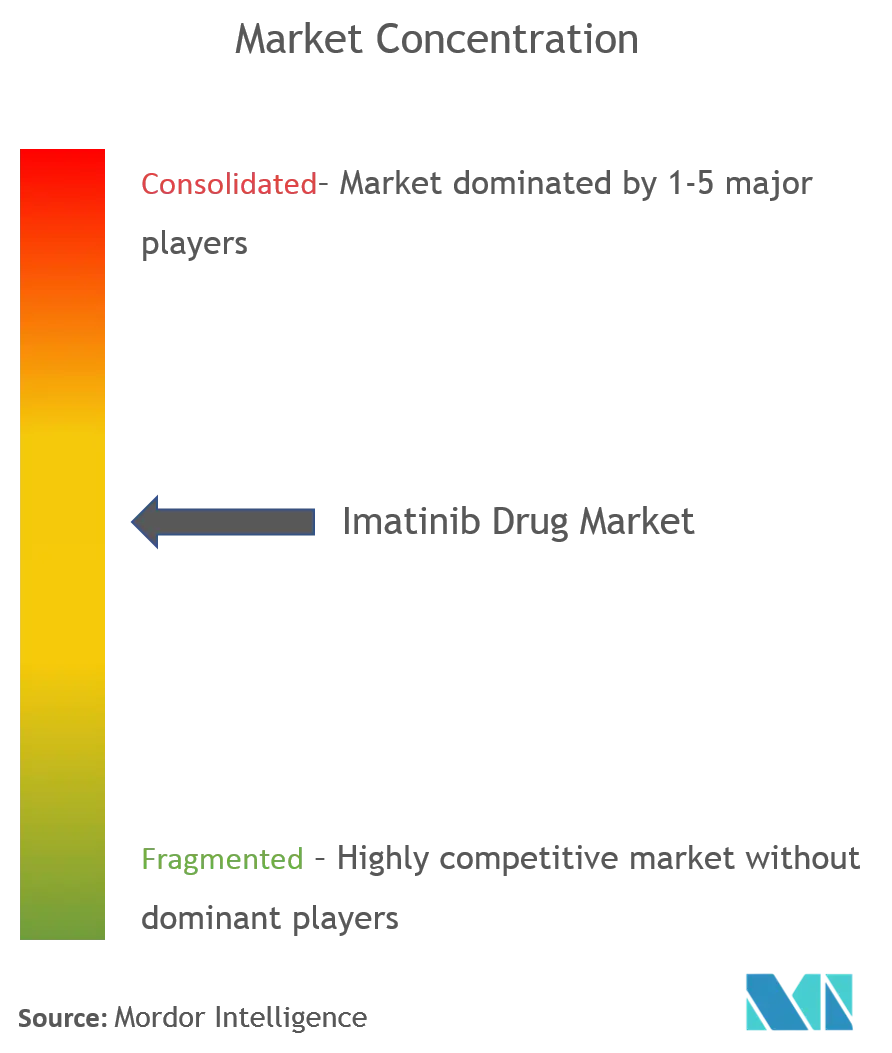
Chronic Myeloid Leukemia Treatment Market Expected to Expand at a Steady 2022-2028 | Teva Pharmaceutical Industries Ltd, F. Hoffmann-La Roche Ltd., Novartis AG - Digital Journal

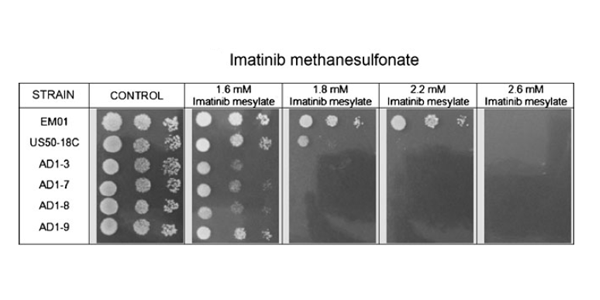
PDF) Efficacy and safety of generic imatinib after switching from original imatinib in patients treated for chronic myeloid leukemia in the United States

These highlights do not include all the information needed to use IMATINIB MESYLATE TABLETS safely and effectively. See full prescribing information for IMATINIB MESYLATE TABLETS. IMATINIB MESYLATE tablets, for oral use Initial

Population pharmacokinetic modelling of imatinib in healthy subjects receiving a single dose of 400 mg | Cancer Chemotherapy and Pharmacology
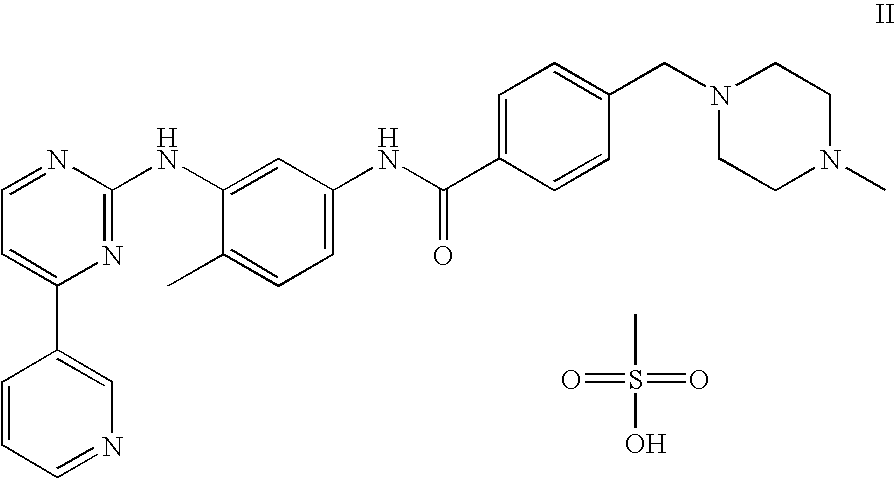
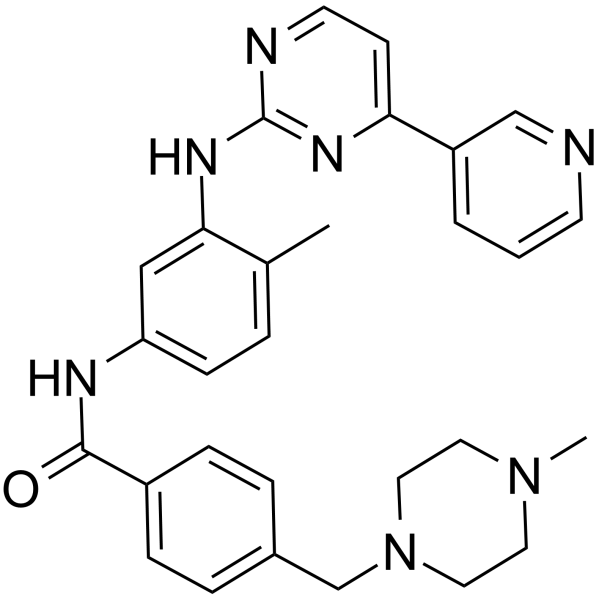
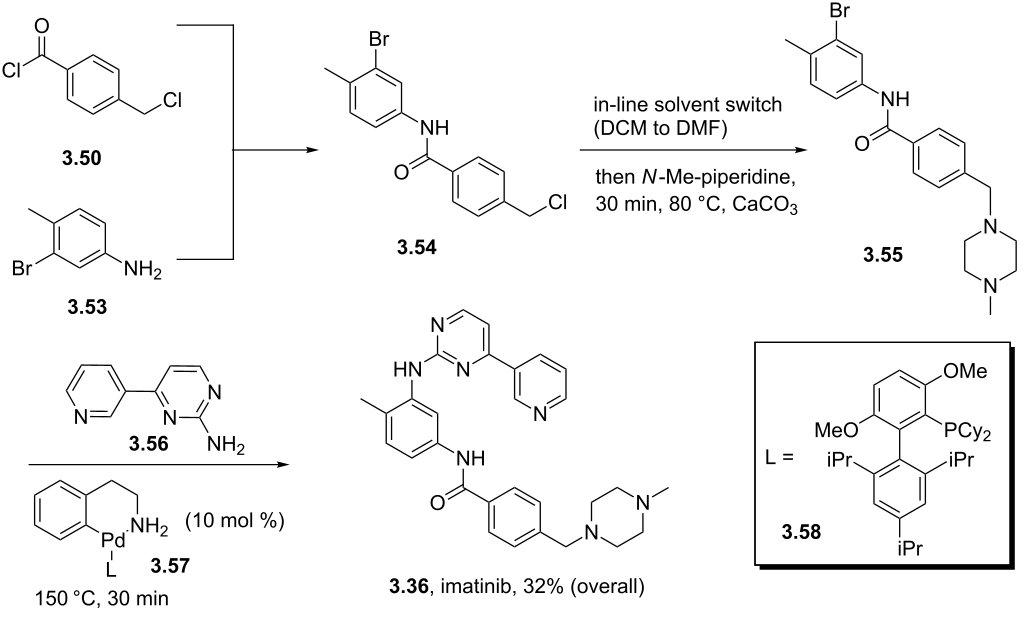
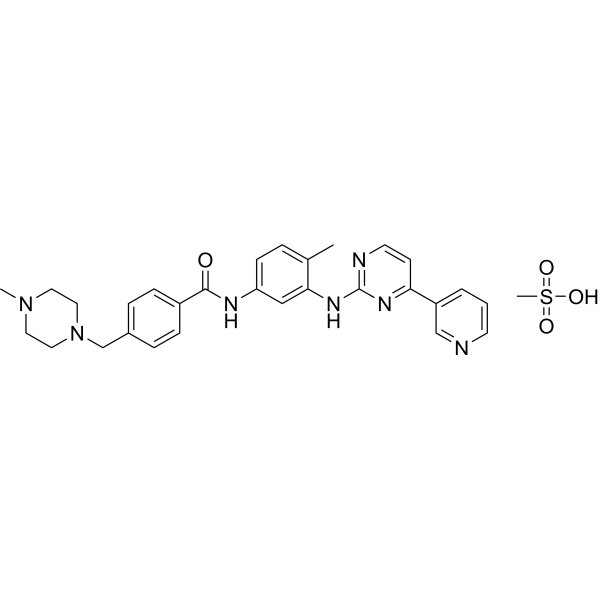
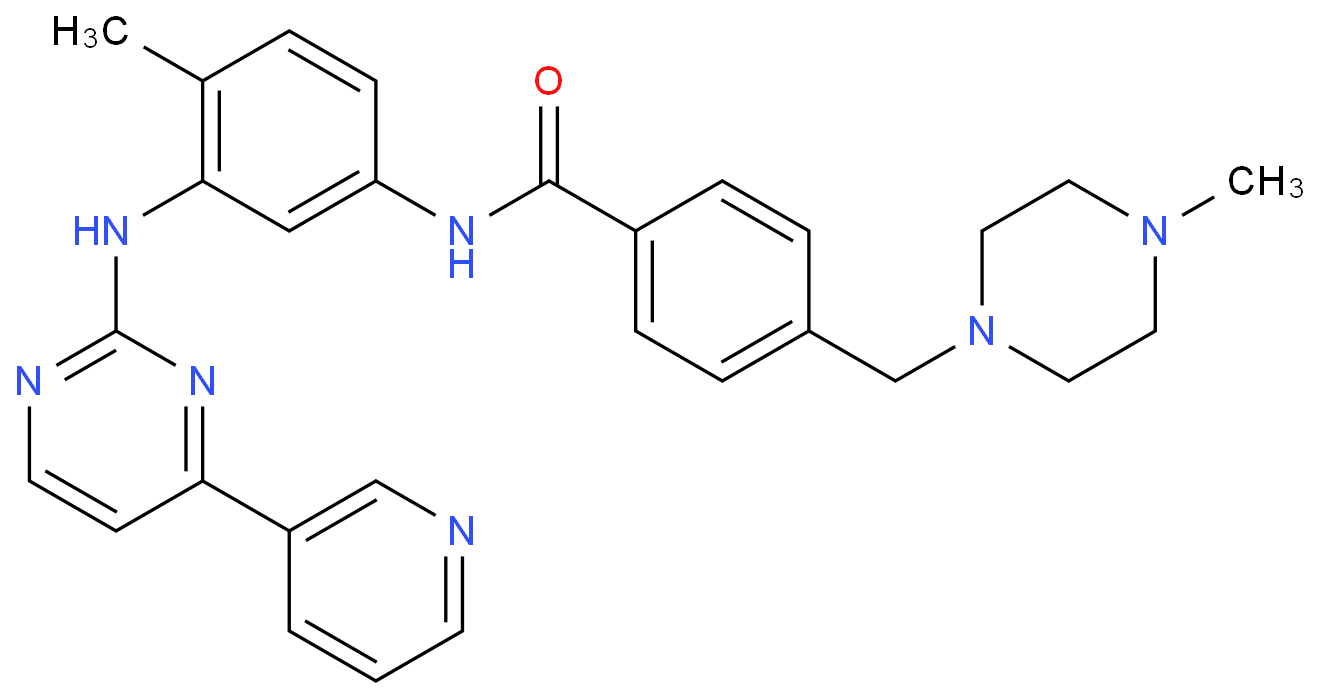
US8269003B2 - Stable crystal form of imatinib mesylate and process for the preparation thereof - Google Patents
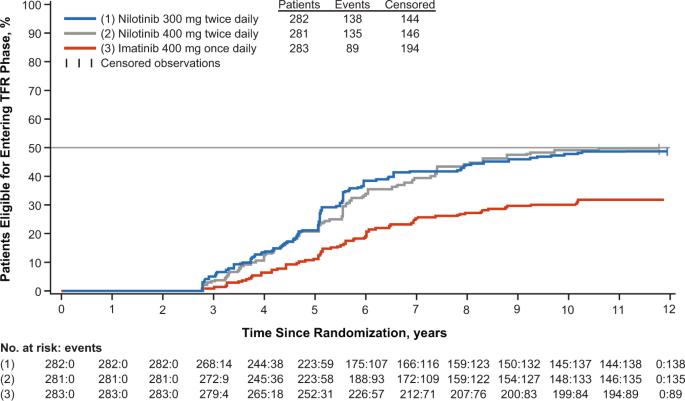
Long-term outcomes with frontline nilotinib versus imatinib in newly diagnosed chronic myeloid leukemia in chronic phase: ENESTnd 10-year analysis | Leukemia

OMACETAXINE MEPESUCCINATE SHOWS ACTIVITY IN CHRONIC MYELOID LEUKEMIA PATIENTS WHO HAVE BECOME RESISTANT TO OR INTOLERANT OF PRIOR TYROSINE KINASE INHIBITORS (TKIs) (1,2,3)